21 CFR Part 11 & EU GMP Annex 11 Compliance
Ensure global regulatory compliance with digital signature workflows that meet FDA and EU electronic records standards for pharmaceuticals and biotech operations.
Pharma & Biotech Contract Management Solution
21 CFR Part 11 Compliant, CSV-Validated, ALCOA+ Ready Electronic Signatures, Full Audit Trails, Faster, Safer for Workflows.
Start Your Free Trial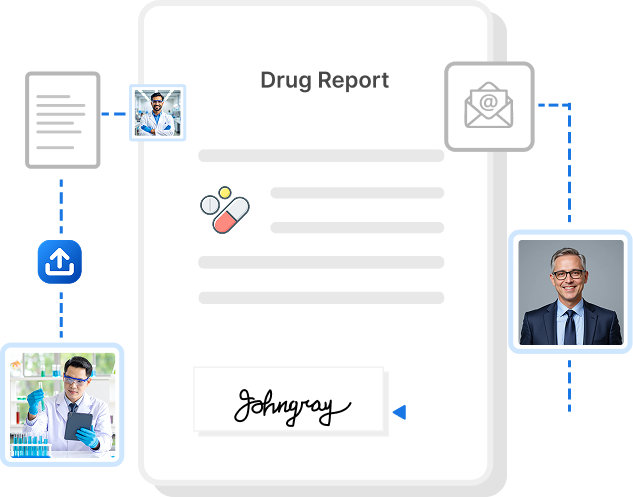














Per Week Saved

Fast Signature Approval

21 CFR Part 11 Compliant
Ensure global regulatory compliance with digital signature workflows that meet FDA and EU electronic records standards for pharmaceuticals and biotech operations.

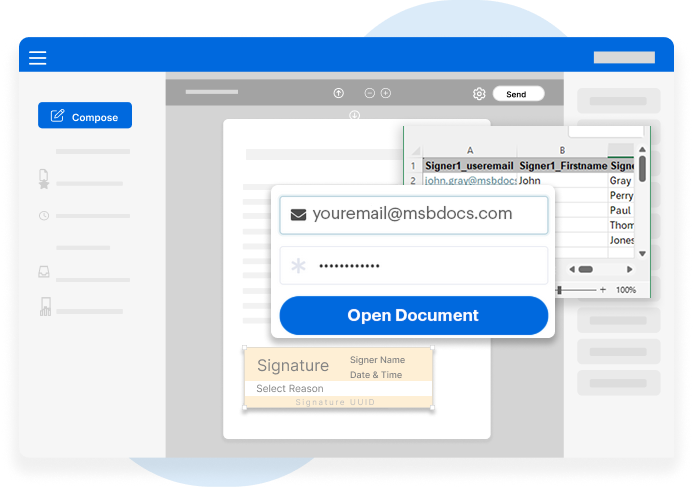
MSB Docs is fully CSV-ready, helping you meet strict validation requirements for systems that manage GxP-relevant data and electronic records.
Maintain data accuracy and reliability with built-in adherence to ALCOA+ principles, Attributable, Legible, Contemporaneous, Original, Accurate, and more.
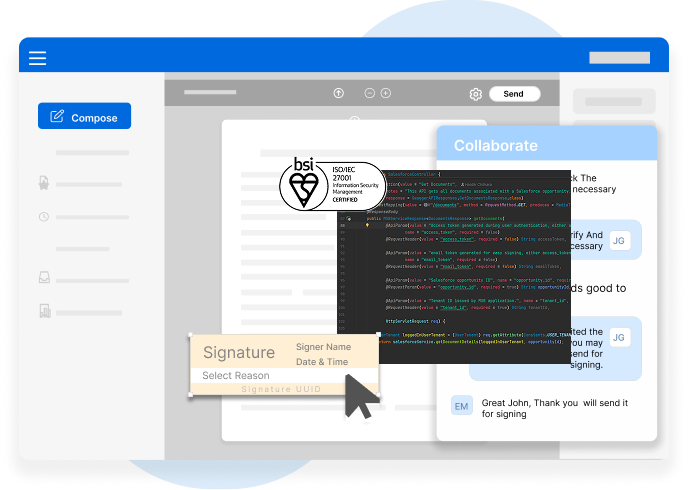
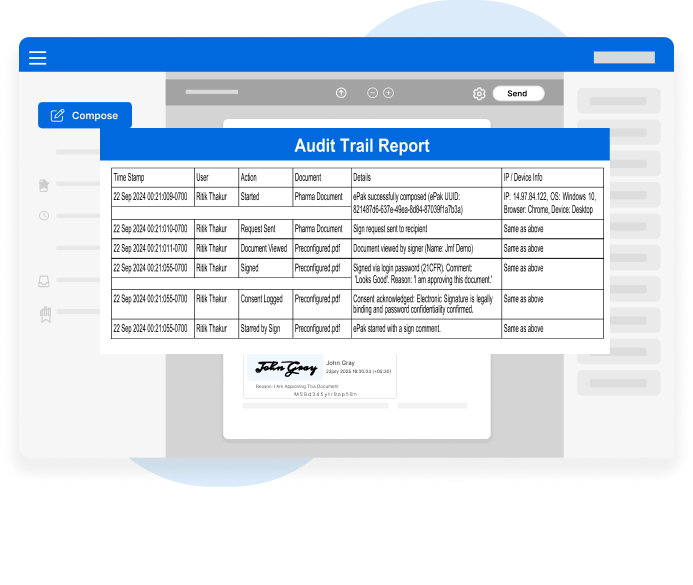
Every action is time-stamped, traceable, and tamper-evident ensuring readiness for inspections, audits, and internal quality checks.
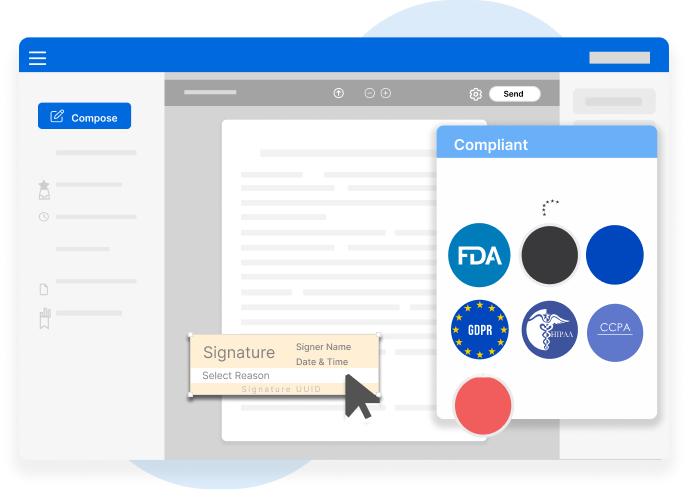
Secure, jurisdiction-compliant digital signatures that meet global standards including FDA, eIDAS, IT Act 2000, GDPR, HIPAA, CCPA, and DPF.
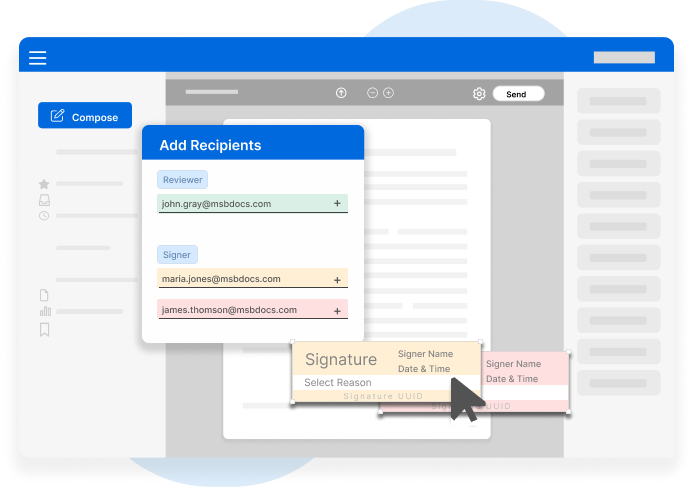
Control access based on user roles, departments, or document type. Ensure that only authorized personnel can view, sign, or modify sensitive data.
MSB Docs protects the integrity and authenticity of electronic records in full compliance with global standards

Digitally sign CVs, GCP training, licenses, and financial disclosures for faster site initiation.
Capture remote patient consent on ICFs and HIPAA forms with compliant, timestamped eSignatures.
Digitize patient onboarding forms, recruitment logs, and screening checklists with secure, real-time tracking.
Digitally sign Form 1572, CTAs, NDAs, and financial agreements with audit-ready compliance.
Sign IRB approvals, lab certifications, site feasibility and delegation log digitally.
eSign deviation reports, CAPAs, and incident logs with traceable, audit-compliant workflows.
Allow CRAs to upload and sign monitoring docs from remote locations securely.
Capture and sign trial data onsite using mobile apps with encrypted sync.
eSign validation protocols, deviation logs, and change controls to meet GxP and data integrity standards.
Apply compliant eSignatures on audit trails, quality records, and electronic batch documentation.
Apply compliant eSignatures on audit trails, quality records, and electronic batch documentation.
Digitally sign IQ/OQ/PQ protocols, risk assessments, and system validation records.
eSign CAPA plans, response letters, and supporting documents for post-483 observations.
Approve and sign cGMP audit findings, internal inspection reports, and follow-up actions.
Digitally execute forms like IND, NDA, ANDA, and Module 1 submission letters.
eSign system audit trails, validation summaries, and deviation tracking logs securely.
Collect electronic signatures for SOP approvals, training acknowledgements, and version updates.
Digitally sign audit reports, compliance certificates, and lab certification documents for regulatory readiness.
eSign test results, analytical data sheets, and COAs to ensure traceable QC documentation.
Approve batch records, release forms, and QA disposition reports with compliant digital signatures.
Sign off calibration certificates, maintenance logs, and verification checklists electronically.
eSign temperature, humidity, and contamination logs to maintain cGMP-compliant facility conditions.
Digitally authorize manufacturing start-up checklists, run sheets, and production logs.
Apply eSignatures to IPQA checklists, sampling records, and deviation observations in real time.
eSign MSDS forms, material transfer records, and hazardous material handling logs securely.
eSign CRO contracts, SOWs, and collaboration terms to accelerate clinical outsourcing workflows.
Digitally execute supply agreements, SLAs, and quality agreements for faster procurement cycles.
Streamline MSAs and addendums with traceable eSignatures and automated approval workflows.
eSign mutual and one-way NDAs with tamper-proof audit trails and signer authentication.
Digitally sign technology transfer, co-development, and patent licensing agreements securely.
eSign regional distribution contracts and channel partner agreements with real-time tracking.
Apply eSignatures on inter-institutional data sharing and clinical data access agreements.
Execute grant agreements, payment terms, and budget approvals for investigator sites digitally.
eSign attendance logs, trainer sign-offs, and training completion forms for audit readiness.
Digitally capture signatures on online module completions and assessment acknowledgements.
eSign compliance training certificates and competency validation forms for regulatory audits.
Digitally execute NDAs, offer letters, tax forms, and role acceptance documents.
Capture eSignatures on CE program participation, completion, and re-certification logs.
eSign access approvals, system role assignment forms, and permission change logs.
Digitally sign appraisal forms, development plans, and workplace policy acknowledgements.
eSign PAP applications, eligibility declarations, and consent forms to expedite access.
Digitally execute physician requests, approvals, and patient consent for unapproved therapies.
eSign prior authorizations, payer forms, and benefit verification documents securely.
Capture eSignatures on patient intake, opt-in, and support program enrollment documents.
Digitally sign service logs, escalation notes, and issue resolution confirmations.
eSign rep visit summaries, HCP interaction records, and sample drop-off forms on mobile.
Apply eSignatures to escalation approvals, issue reports, and case closure confirmations.
Compliment your current business development with Unconstrained predictive analytics.